 Formal chargeC 4 (1/2) × 8 0 0 This molecule is neutral. Check when youll get cost of living payments. In this video you will learn how to quickly calculate formal charges. Your payment will change each month if you earn a different amount, or if your situation changes.  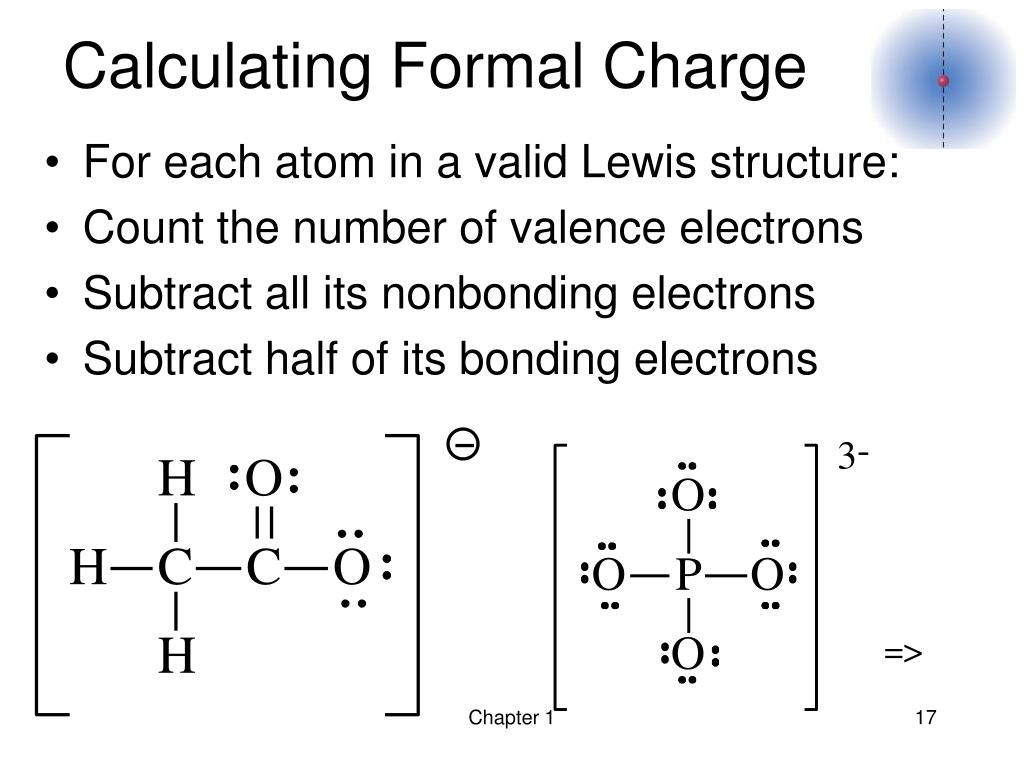
Yes, just switch the single and double bonds for the above two structures (with the oxygens and their lone electrons). Formal chargeH 1 (1/2) × 2 0 0 This applies to each hydrogen. The easy way to do this is to compare the number of bonds each atom has with the number. In the left resonance structure, all the atoms have zero formal charge, while on the right structure, the nitrogen has a +1 formal charge, and the oxygen with the single bond has a -1 formal charge.Ĭan you draw two additional resonance structures for the above molecules? What is the Formal Charge of an Atom Anytime you see a charge in a molecule, it tells you that the atom with the charge has more or less electrons than it. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
